Science > Physics > Electrostatics > Introduction to Static Electricity
Electricity is a very important form of energy which can be easily converted into other forms of energy. Electricity can be produced at one place and can be transmitted to long distances. Electricity is a branch of Physics which deals with charges, stationary and moving. For convenience, electricity is divided into two types: static electricity and current electricity. The pioneers of this branch Physics are Gilbert, Thale, Faraday, Benjamin Franklin, Ampere, Volta, Coulomb, Thevenin, and Maxwell.
Structure of Atom:
An atom consists of positively charged protons, negatively charged electrons, and neutral neutrons. The total number of protons in an atom is equal to the total number of electrons. Thus net positive charge balances the net negative charge. Hence atom is electrically neutral.
Protons and neutrons are present in central core called nucleus. Hence nucleus carries a positive charge. The negatively charged electrons revolve around the positively charged nucleus in circular orbits. There is force attraction between the negatively charged electrons and the positively charged nucleus which provides the necessary centripetal force for the circular motion of electrons around the nucleus.
The attractive force between the electron and the nucleus decreases with the increase in the distance of the electron from the centre of the nucleus. Thus the electrons present in the last orbit and last subshells (orbitals) are loosely attached to the nucleus. These orbitals are called valence orbitals and such electrons are called valence electrons,
By applying suitable method, these valence electrons are removed from valence orbitals and made to move in a particular direction or can be made to transfer to another substance.
Frictional Electricity:
Thale’s Experiment:
Around 600 BC (Before Christ), a Greek mathematician Thales discovered that amber (a resinous material) rubbed with animal fur attracted light objects like pieces paper, feathers, and treads. Both the amber and the fur acquired this property of attracting lighter objects. Amber in the Greek language is called ‘electron’. From this word, Thale coined a word ‘electricity’. Even though other people may have noticed this before, Thales was the first to record his findings. We don’t have his writings, but from other people’s reports of his work, we can guess at his experiments. At this time, magnetism was also confused with static electricity. In this experiment, it is said that the amber and fur have acquired electrical property and process of acquiring electrical property is called electrification.
Explanation:
When a body is rubbed over another, the transfer of valence electrons from one substance to another takes place. The body which loses valence electrons become electron deficient and acquires a positive charge, while the body which gets the electrons becomes electron rich and acquires a negative charge. As the electricity is produced by rubbing (friction), this electricity is called as frictional electricity.
A charge may be defined as the amount of electricity present in a body. S.I. unit of charge is coulomb (C) named after Charles Coulomb. It is a scalar quantity and its dimensions are [LoMoA1T1].
Gilbert’s Experiment:
In the early sixteenth century, Gilbert performed the similar example as Thales had performed but he used other materials like a glass rod, ebonite rod etc. He also gave characteristics of charges developed on the bodies.
When a glass rod is rubbed with a silk cloth, the loosely attached valence electrons of glass rod get transferred to the silk cloth. Thus in case of glass rod becomes electron deficient and acquires a positive charge, while the silk cloth has the excess of negative charge and acquires a negative charge. The total charge of the system i.e. the glass rod and the silk cloth remains zero.
When an ebonite rod is rubbed with a fur, the loosely attached valence electrons of fur get transferred to the ebonite rod. Thus ebonite rod has e the cess of electrons and acquires a negative charge, while the fur is electron deficient and acquires a positive charge. The total charge of the system i.e. the ebonite rod and the fur remains zero.
Gilbert charged two glass rods and found that the two glass rods repel each other. Then he charged two ebonite rods and found that the two ebonite rods repel each other. From this, he concluded that the like charges repel each other. He charged glass rod and bought a charged ebonite rod near to glass rod, he found that the two rods attract each other. From this, he concluded that the unlike charges attract each other.
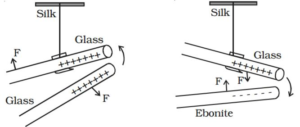
Thus there are two types of charges positive and negative. Charge produced on glass rod by rubbing it with a silk cloth is considered as a positive charge, while charge produced on ebonite is considered as a negative charge.
For electrification, two material bodies are involved. The following list gives some objects which are arranged in the order such that if the two objects from the list are rubbed together, they get electrified. The object appearing first in the list acquires a positive charge and the object which appears later in the list acquires a negative charge.
| 1. Cat’s skin | 2. Fur | 3. glass | 4. Cotton |
| 5. Silk | 6. Wood | 7. Indian rubber | 8. Resin |
| 9. Amber | 10. sulphur | 11. ebonite |
Characteristics of Charges:
- There are two types of charges, namely positive and negative.
- Like charges ( positive and positive or negative and negative) repel each other. Unlike charges (positive and negative) attract each other.
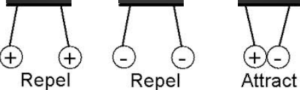
- When neutral body accepts excess of electrons then it acquires a negative charge, while a neutral body loses electrons, then it acquires a positive charge.
- The total charge on a body (either positive or negative) is an integral multiple of the magnitude of the charge on an electron.
- When a glass rod is rubbed with a silk cloth, the glass rod acquires a positive charge and the silk cloth acquires negative charge, but the total charge of the system i.e. combined charge on a glass rod and silk cloth is zero.

- The charge per unit surface area is called surface charge density. It is more at sharp curves and pointed tips. The charge always remains on the outer surface of a conductor. On the uniform surface, it gets distributed uniformly.

- If the charge is produced on insulator, then it remains stationary at the supplied position on the insulator, while if the charge is produced on a conductor, then it gets distributed on the surface of the conductor.
- The charge supplied to a conductor always reside on the outer surface.
- Electrical charge is a scalar quantity.
Experiments to Study Characteristics of Electrical Charges:
Coulomb’s or Biot’s Experiment:
Coulomb demonstrated that charge always resides on the outside surface of a conductor with the aid of two hemispherical cups called Coulomb’s hemispheres which fitted exactly round an insulated metal sphere. These Coulomb’s hemispheres have insulating holders attached to them.

The sphere mounted on the insulated stand is first charged, and afterwards, the hemispheres are fitted over it by holding at insulating handles. On removing the hemispheres they are found to be charged, but the sphere becomes chargeless. This shows that all the charge on the sphere must have passed to the outside of the hemispheres.
Faraday’s Butterfly Net Experiment:
Michael Faraday used an insulated cotton net to act as a hollow conductor. at cone apex of the net, a silk thread is attached which extends on both the sides, pulling which the net surfaces can be turned inside out.

The charge is given to inside surface of the net and it is observed that the charge gets distributed on the outer surface of the net.
Now the string is pulled from the ringside so that the surfaces flip. i.e. the inside surface becomes the outside surface and the outside surface becomes the inside surface. Again charge is found residing on the outer surface.
Atmospheric Electricity:
Benjamin Franklin discovered atmospheric electricity. When he was flying a kite made up of silk cloth and iron wire as the cross of the kite on a rainy day, he got electricity shocks through the silk thread connected to the kite. From this, he concluded that the clouds contain electric charges.
Reason for the Charge on Clouds:
The heat from the sun and hot air causes evaporation of water bodies like the sea, river, etc. to vapourize. These vapours rise to height and get accumulated in the sky. This accumulation of vapour particles is the cause of formation of clouds.
When the molecules of cloud rub with each other, and thus they get charged due to friction. A large amount of electrostatic charge (either positive or negative) gets accumulated on the clouds, resulting in the increase in its electrical potential.
When two clouds of high electrical potential but carrying opposite charges come to each other, the air between them gets ionized and the electrons jump from negatively charged cloud to positively charged cloud. It produces a dazzling white streak of light called the lightning.
During lightning, a large amount of heat is produced, which creates pressure waves, which are transferred in all the directions, and produce a very loud sound called the thunder.
Sometimes during lightning, the accumulated charge on clouds tends to conduct to the surface of the earth. It is to be noted that the charge ties to strikes a conductor that is very near to it like tall buildings and trees. A negatively charged cloud induces a positive charge at the top of the building and a negative charge at its base. Thus a potential difference is created between the top and base of the building. During the rain, the moist air creates a path between the cloud and the top of the building creating a path for the flow of the charge. When the electrons from the cloud reach the top of the building a very high current is set between the top of the building towards the base, which may cause a fire to the building. Thus lightning can have a very devastating effect. To protect high rise buildings from this destruction a lightning rod is put at the top of the building which is connected to the earth. Thus now the electrical current has the easy path through the lightning rod. When a charge is received it is passed to the ground through the lightning rod and not through building itself. Thus the building is safeguarded from the devastating effect of the lightning.


2 replies on “Introduction to Static Electricity”
Superb🔥 explanation
Thank you
Concepts explained clearly with diagrams , thanks