Science > Chemistry > Physical Chemistry > Ionic Equilibria > Common Ion Effect
In this article, we shall study the common ion effect and its applications.
The phenomenon in which the degree of dissociation of any weak electrolyte is suppressed by adding a small amount of strong electrolyte containing a common ion is called a common ion effect.
Example – 1: (Dissociation of a Weak Acid)
Ionization of weak electrolyte acetic acid (CH3COOH) is suppressed by adding strong electrolyte sodium acetate (CH3COONa) containing common acetate ion (CH3COO–)
Explanation:
Suppose, an electrolyte acetic acid (CH3COOH) is treated with water. It dissociates and an equilibrium exists as follows,
CH3COOH(aq) ⇌ CH3COO–(aq) + H+(aq)
By applying the law of mass action,
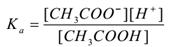
Where ‘Ka’ is the dissociation constant of acid.
If a small amount of a strong electrolyte like sodium acetate (CH3COONa) is added to the aqueous solution of CH3COOH, it gets dissociated and equilibrium exists, as
CH3COONa(aq) → CH3COO–(aq) + Na+(aq)
Here CH3COO– ions are common hence their concentration increases. According to Le-Chatelier’s principle, equilibrium shifts towards the left. To keep the value of Ka constant, the concentration of CH3COOH molecules is increased. In this way ionisation of CH3COOH is suppressed by adding CH3COONa. Thus the pH of the solution increases.
Example – 2: (Dissociation of a Weak Base):
Ionisation of weak electrolyte Ammonium hydroxide (NH4OH) is suppressed by adding strong electrolyte Ammonium chloride (NH4Cl) containing common ammonium ion (NH4+)
Explanation:
Suppose, an electrolyte Ammonium hydroxide (NH4OH) is treated with water. It dissociates and an equilibrium exists as follows,
NH4OH(aq) ⇌ NH4+(aq) + OH–(aq)
By applying the law of mass action,
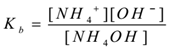
Where ‘Kb’ is dissociation constant of the base.
If a small amount of a strong electrolyte like Ammonium chloride (NH4Cl) is added to the aqueous solution of NH4OH, it gets dissociated and equilibrium exists, as
NH4Cl(aq) → NH4+(aq) + Cl–(aq)
Here NH4+ ions are common hence their concentration increases. According to Le-Chatelier’s principle, equilibrium shifts towards the left. To keep the value of Kb constant, the concentration of NH4OH molecules is increased. In this way ionisation of NH4OH is suppressed by adding NH4Cl. Thus the pH of the solution decreases.
Applications of Common Ion Effect:
Purification of Common Salt:
Principle:
The addition of common ion to a saturated solution of salt causes the precipitation of salt. When the ionic product exceeds the solubility product, precipitation takes place.
Process and Explanation:
A saturated solution of common salt, free from suspended impurities is taken and HCl gas is passed through it. In a saturated solution of impure NaCI, equilibrium exists as follows,
NaCl(aq) → Na+(aq) + Cl–(aq)
If pure HCI gas is passed through this solution, being strong electrolyte, it dissociates almost completely and equilibrium exists,
HCl(aq) → H+(aq) + Cl–(aq)
Now Cl– is a common ion. The concentration of CI– ions (common ions) is increased. According to Le-Chatelier’s principle equilibrium shifts towards the left. Dissociation of NaCI is suppressed and pure NaCI is precipitated as solid salt. Thus pure NaCI can be precipitated by passing HCI gas in the saturated solution of impure NaCI.
Salting Out of Soap:
Principle:
The addition of common ion to a saturated solution of salt causes the precipitation of salt. When the ionic product exceeds the solubility product, precipitation takes place.
Process and Explanation:
Soap is sodium salt of higher fatty acids (RCOONa). It is prepared by hydrolysis of oils with NaOH. In saturated soap solution there exist an equilibrium,
ROONa(aq) → RCOO–(aq) + Na+(aq)
If a small amount of NaCl is added to the saturated soap solution, it dissociated as
NaCl(aq) → Na+(aq) + Cl–(aq)
Concentration of Na+ ions (common ion) increases. Due to the common ion effect, dissociation of soap is decreased and soap gets precipitated and then can be easily removed from the soap solution. This process of getting solid soap from soap solution, by adding salt like NaCI is called salting out of soap.
Washing of precipitate:
Principle:
a) Solubility of a salt is less in a solvent containing a common ion.
Process and Explanation:
Washing of the precipitate means removing impurities from precipitate by suitable liquid / solvent. If a precipitate is washed with water, a part of the precipitate may go into solution. Hence in gravimetric analysis, the precipitate is washed with washing solution containing little strong electrolyte having common ion. Due to common ion effect dissociation of precipitate in washing solution is suppressed. Precipitate is washed without dissolving in washing solution.
Examples:
- Precipitate of BaSO4 is washed with water containing little H2SO4 because of SO4— ions (common ions), the solubility of BaSO4 precipitate is decreased and there is no loss of precipitate during washing.
- Cus precipitate is washed with dilute H2S solution.
- ZnS precipitate is washed with dilute H2S solution
- PbCl2 precipitate is washed with dilute HCl solution.
2 replies on “Common Ion Effect”
Really helpful
Good explanation