Science > Chemistry > Physical Chemistry > Nature of Chemical Bond > Overlapping of Orbitals
In this article, we shall study the formation of bonds by overlapping of orbitals and different types of overlaps.
Formation of Hydrogen Molecule on The Basis Of Orbital Overlap:
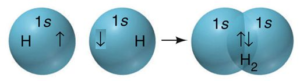
The electronic configuration of a hydrogen atom is 1s1. It contains one unpaired electron in its valence shell. Therefore 1s orbital in the hydrogen atom is bonding orbital. When the two hydrogen atoms having valence electrons with opposite spins approach each other, the attractive force dominates the repulsive force between the electrons in the initial stage. Thus, as the distance between two hydrogen atoms decreases the potential energy of the system gradually decreases.
A state of minimum potential energy is reached when the forces of attraction are balanced by the forces of repulsion between two hydrogen atoms. At this point the energy of the system is minimum and the stability is maximum. At this stage orbital of two hydrogen atoms, overlap and spins of electrons are neutralized and a stable covalent bond is formed between hydrogen atoms, forming H2, molecule. In this case, the overlap of orbitals is maximum. This overlap is called s-s overlap and bond formed are coaxial overlapping of two s orbital is called s-s sigma (σ) bond.
In the formation of a molecule, the release of energy comes from two sources. The neutralization of spin magnetic moments of the two electrons and the accumulation of electron density between two nuclei.
Diagram :
1 S Orbital 1 S Orbital S-S overlap
Reason: Helium does not form a diatomic molecule:
Helium with atomic number 2 has electron configuration 1s2. Thus helium contains two paired electrons in its 1s orbital. The pairing of electrons in 1s orbital neutralizes the spin magnetic moments of each other. This gives stability to the helium atom. There are no empty orbitals in the first shell for unpairing and promoting these paired electrons to higher energy orbital. Thus helium has no unpaired electron i.e. no bonding orbital. Therefore according to valence bond theory, it cannot form a covalent bond with another helium atom.
Each helium atom has paired electrons in 1s is orbital. If two such orbitals come close to each other, there is net repulsion between them because repulsive forces are stronger than the attractive forces. If they do so it increases the potential energy of the system and it becomes unstable. Thus overlapping of orbitals cannot take place. Therefore helium cannot form a diatomic molecule. It exists as a monoatomic gas.
Types of Overlap of Atomic Orbitals:
The term overlap refers to the overlap of the atomic orbitals of the two approaching atoms as they enter into the bond formation stage. In the case of s and p orbitals, there can be three types of overlap.
s – s orbital overlap ( formation of H2 molecule):
The mutual overlap between the half-filled s orbitals of two atoms is called s – s overlap and the covalent bond formed is known as sigma (s) bond. e.g. formation of a hydrogen molecule from two hydrogen atoms.
s – orbital is spherical in shape and overlapping takes place to some extent in all directions. Hence s -s bond is non – directional.
Hydrogen (1s1) atom has 1s orbital containing a single electron i.e. it is half-filled. Two such 1s orbitals from the two hydrogen atoms having electrons with opposite spins approach each other, then the potential energy of the system decreases. The two ‘s’ orbitals overlap each other when they acquire minimum potential energy, forming H-H sigma bond or s-s overlap. H-H bond is a nonpolar covalent bond.
As the two orbitals are overlapping such that the overlapped region lies on the line joining the two nuclei of the overlapping orbitals (axial overlapping) bond formed is sigma bond.
Diagram :

1 S Orbital 1 S Orbital S-S overlap
p – p orbital overlap (Formation of Fluorine F2 molecule):
The mutual overlap between two half-filled p – orbitals of two atoms is called p – p overlap and the covalent bond formed is known as p – p bond. If the overlapping takes place along the internuclear axis the bond is called sigma bond and if the overlapping takes place literally the bond is known as pi bond. e.g. formation of fluorine molecule from two fluorine atoms. CI2, Br2and I2 are also formed by p – p overlap.
The atomic number of fluorine is 9. The electronic configuration of the fluorine atom is 1s2. 2s2. 2px2, 2py2, 2pz1. Each F atom has one unpaired electron in p – orbital (pz orbital). When two fluorine atoms each containing unpaired electron with opposite spins approach each other, then the potential energy of the system decreases. The two ‘p’ orbitals overlap each other when they acquire minimum potential energy.
As the two orbitals are overlapping such that the overlapped region lies on the line joining the two nuclei of the overlapping orbitals (axial overlapping) bond formed is sigma bond. As p orbitals are dumbbell-shaped they overlap in a particular direction. Therefore the p-p bond is directional. The back lobe of the overlapping p orbital is distorted and its size is reduced. This is due to the tendency of the electron to remain in the overlapping region.
F-F bond is a non-polar, as shared pair of electrons is attracted by both the atoms.
Diagram :

1 P Orbital 1 P Orbital P-P overlap
s – p orbital overlap (Formation of Hydrogen Fluoride Molecule):
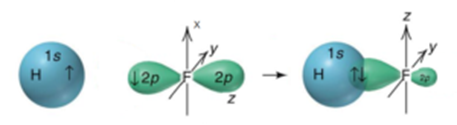
The overlap between the half-filled s – orbital of one atom and the half-filled p – orbital of another atom is called s – p overlap and the covalent bond formed is known as s – p sigma bond. E.g.: Formation of HF molecule, H – X bond in HCI, HBr, and HI are also formed by s-p overlap.
The electronic configuration of a hydrogen atom is 1s1, while that of fluorine atom is 1s2. 2s2. 2px2, 2py2, 2pz1. Thus 1s orbital of a hydrogen atom and the 2p orbital of fluorine atom containing unpaired electron can overlap and the bond formation is possible provided the spin of electrons in overlapping orbitals is opposite. The bond in hydrogen fluoride is a s – p bond. As the two orbitals are overlapping such that the overlapped region lies on the line joining the two nuclei of the overlapping orbitals (axial overlapping) bond formed is sigma bond. The back lobe of the overlapping p orbital is distorted and its size is reduced. This is due to the tendency of the electron to remain in the overlapping region.
Fluorine is more electronegative than hydrogen, hence H-F bond is polar.
Diagram :

Reason: H – F bond is polar.
HF molecule is formed by the s-p overlap of orbitals. On Pauling scale, the electronegativity of H is 2.1 and that of F is 4. Thus fluorine is more electronegative than H, the electronic cloud in this bond is displaced more towards fluorine. In other words, the electron pair shared between the two atoms is attracted more towards fluorine. This gives fluorine atom a partial negative charge (δ -) and hydrogen atom a partial charge (δ +).
Thus the H – F bond is not purely covalent. It possesses a partial ionic character and is said to be a polar bond. The bond has 43% ionic character. H – X bond in HCI, HBr, and HI are also polar since CI, Br and I are more electronegative than H.
4 replies on “Overlapping of Orbitals”
This page contains all the quality to be able to understand this Valence bond theory
Thanks a lot
It’s really helpful and educative
Very concise
thank u