In the last article, we have studied the initial classification of elements. In this article, we shall study the modern periodic table.
Moseley’s Criteria:
Henry Moseley, a physicist from England in the year 1913, observed that when certain metals were bombarded with high-speed electrons, X-rays are emitted. He studied the frequencies of the X-rays emitted and found that in all the cases, the square root of the frequency was directly proportional to the atomic number of the atom of the metal.
He plotted a graph between the square root of frequency and atomic numbers of different metals, a straight line was obtained. When a graph was plotted between the square root of frequency and atomic masses of the metals, the graph was not a straight line.
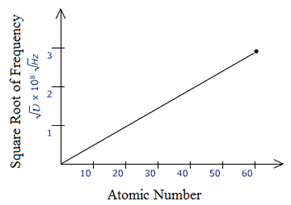
He concluded that there was no correlation between the frequency and the atomic mass. From these observations, Moseley concluded that atomic number and not the atomic mass is, the fundamental property of an element.
On the basis of this hypothesis, he put forward his law of periodicity, commonly known as Modern Periodic Law. It states that “The physical and chemical properties of the elements are the periodic function of their atomic numbers”. It is the basis of “Modern Periodic Table”. Thus Moseley modified Mendeleev’s periodic law.
Explanation of the Modern Periodic Law on the Basis of Electronic Configuration:
In chemical reactions, there is a transfer or sharing of electrons. Thus chemical properties of an element are dependent on the electronic configuration (number of electrons). In an atom, the number of electrons depends on the number of protons (they are equal). The number of protons in an atom depends on the atomic number of the element.
Similarly, physical properties of an element are dependent on the configuration of electrons in an atom of the element. Hence the physical and chemical properties of an element are the function of atomic number.
Periodicity and Magic Numbers:
Periodicity may be defined as the repetition of the similar properties of the elements placed in a group and separated by certain definite gaps of atomic numbers.
When the elements were arranged in increasing order of atomic numbers, it was observed that the properties of elements were repeated after certain regular intervals 01 2, 8, 8, 18, 18 and 32. These numbers are called magic numbers. These numbers are the cause of periodicity in properties due to the repetition of similar electronic configuration.
Features of Modern Periodic Table:
Modern Periodic Table is based on the Modern Periodic Law which states that “The physical and chemical properties of the elements are the periodic function of their atomic numbers”. The elements are arranged in order of increasing atomic numbers in horizontal rows called periods and vertical columns called groups.
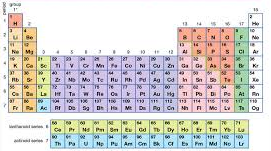
Groups:
- There are eighteen (18) groups in the Modern Periodic Table. These are numbered from 1 to 18.
- The elements present in a group are separated by definite gaps of atomic numbers (8, 8, 18, 18, 32).
- The elements present in a group have the same number of electrons in the valence shell of their atoms. i.e. their electronic configuration of valence shell is identical. For example, all the elements of group 1 have valence shell configuration of ns1. All the elements of group 17 have valence shell configuration of ns2np5.
- Due to similar valence shell configuration, the elements present in a group have identical chemical properties.
- The physical properties of the elements in a group such as melting point, boiling point, density vary gradually.
- Elements of groups, 1, 2, 13, 14, 15, 16, and 17 are called normal or representative elements.
- Elements from Gr. 3 to 12 are called transition elements.
- Noble or inert gases are placed in group 18.
- Two series of fourteen elements each are called at bottom of the periodic table. The elements of these series are called Lanthanoids and Actinoids. These elements are also called inner transition elements or rare earth elements.
- Lanthanoids are placed in group 3 and the sixth period.
- Actinoids are placed in group 3 and the seventh period.
- Elements of group 1are called alkali metals.
- Elements of group 2 are called alkaline earth metals.
- Elements of group 16 are called chalcogens [ore forming elements].
- Elements of group 17 are called halogens. [sea salt forming]
- Elements of group 18 are called noble gases.
Periods:
- Modern Periodic Table has seven (7) horizontal rows called periods. They are numbered 1-7.
- The first (1H – 2He) period has two elements and is called the shortest period.
- The second (3Li – 10Ne) & third (11Na – 18Ar) periods have eight elements and are called short periods.
- The fourth (19K – 36Kr) and the fifth (37Rb – 54Xe) periods have eighteen elements and are called long periods.
- The sixth (55Cs – 86Rn) period contains 32 elements and is called the longest period.
- The seventh (87Fr – )period has 23 elements and is called an incomplete period.
- In all the elements present in a period, the electrons are filled in the same valence shell.
- As the number of electrons in the valence shell change, the chemical properties of the elements present in a period also change.
Merits of the Modern Periodic Table:
- It is quite easy to remember and reproduce.
- The positions of the elements in the periodic table are linked with their electronic configuration.
- Each group is an independent group and the idea of sub-groups as in the case of Mendeleev’s periodic table has been discarded.
- There is one position for all the isotopes of an element because the isotopes have the same atomic number. For example, the three isotopes of hydrogen i.e. protium, deuterium, and tritium have different atomic mass numbers but have the same atomic number one.
- The positions of certain elements which were earlier misfits in Mendeleev’s periodic table are now justified because it is based on an atomic number of the elements.
- This periodic table is linked with the electronic configuration. Hence the further division of the elements into s, p, d, and f blocks has been quite useful in understanding their properties.
Limitations of Long Form of Periodic Table:
- In the long form of the Periodic Table, the position of hydrogen still remains uncertain.
- The inner-transition elements do not find a place in the main body of the table. They are placed separately below the periodic table.
2 replies on “Modern Periodic Table”
I like it
Best information dia
Thanks a lot