Science > Chemistry > Solid State > Electrical Properties of Solids
In the last article, we have studied the dielectric properties of solids. In this article, we shall study the electrical properties of solids.
Electrical Conductivity:
The electrical conductivity of solids is due to the motion of electron or positive holes. The conductivity due to the motion of electron or positive holes is called electronic conductivity. The electrical conductivity may be due to the motion of ions. The conductivity due to the motion of ions is called ionic conductivity. Conductivity due to electrons is called n-type conductivity while that due to holes is called p-type conductivity.
In metals, electrical conductivity is due to the motion of electrons and the electrical conductivity increases with the increase in the electrons available for conduction, the electrical conductivity increases. In pure ionic solids, ions are not available for conduction hence in the pure solid state they are insulators. Due to the presence of defects in the crystal electrical conductivity increases.
Solids exhibit a varying range of electrical conductivities, extending of magnitude ranging from 10–20 to 107 ohm–1 m–1. Solids can be classified into three types on the basis of their conductivities. The difference in conductivities of conductors, insulators, and semiconductors can be explained on the basis of band theory.
Electrical Conductivity on the Basis of Energy Bands:
The group of discrete but closely spaced energy levels for the orbital electrons in a particular orbit is called energy band. Inside the crystal, each electron has a unique position and no two electrons see exactly the same pattern of surrounding charges. Because of this, each electron will have a different energy level. These different energy levels with continuous energy variation form what are called energy bands.
The energy band which includes the energy levels of the valence electrons is called the valence band. The energy band above the valence band is called the conduction band. With no external energy, all the valence electrons will reside in the valence band.
On the basis of energy bands and electrical conductivity, solids can be classified as:
Conductors:
The solids with conductivities ranging between 104 to 107 ohm–1 m–1 are called conductors. Metals have conductivities in the order of 107 ohm–1 m–1 are good conductors. In conductors, the lowest level in the conduction band happens to be lower than the highest level of the valence band and hence the conduction band and the valence band overlap. Hence the electron in the valence band can migrate very easily into the conduction band. Thus at room temperature, a large number of electrons are available for conduction. Examples: Copper, Aluminium, Silver, Gold, All metals
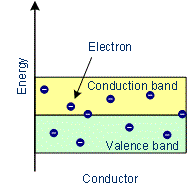
Characteristics of Conductors:
- The substances which conduct electricity through them to a greater extent are called conductors.
- In conductors, the conduction band and valence band overlap with each other or the gap between them is very small.
- There are free electrons in the conduction band.
- Due to increase in temperature conductance decreases.
- There is no effect of the addition of impurities on the conductivity of conductors.
- Their conductivity ranges between 104 to 107 ohm–1 m–1.
Conduction in Metallic Solids:
A metal conductor conducts electricity through the movement of free electrons. Metals conduct electricity in solid as well as a molten state. The conduction of electricity is due to the transfer of electrons and not due to the transfer of matter. The conductivity of metals depends upon the number of valence electrons available per atom. It is nearly independent of the presence of impurity and lattice defects. Conductivity decreases with the increase in the temperature. It can be explained as follows
M → Mn+kernel + ne– free electrons
The kernels are fixed. Due to the increase in temperature, the amplitude of vibration of kernels increases. Hence the obstruction to the flow of electron increases.
Insulators :
These are the solids with very low conductivities ranging between 10–20 to 10–10 ohm–1 m–1. The conduction band and valence band are widely spaced. Thus forbidden energy gap between the valence band and conduction band is large (greater than 3 eV). Hence the electron in the valence band cannot migrate into the conduction band. Hence no electrons are available for conduction. But at a higher temperature, some of the electrons from the valence band may gain external energy to cross the gap between the conduction band and the valence band. Then these electrons will move into the conduction band. At the same time, they will create vacant energy levels in the valence band where other valence electrons can move. Thus the process creates the possibility of conduction due to electrons in conduction band as well as due to vacancies in the valence band. Examples: Glass, wood, paper, plastic, mica.
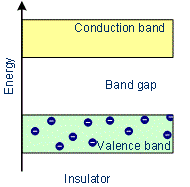
Characteristics of Insulators:
- In Insulators the conduction band and valence band are widely separated.
- There are no free electrons in the conduction band.
- There is an energy gap between the conduction band and the valence band which is more than 3 eV.
- There is no effect of change of temperature on the conductivity of insulators.
- There is no effect of the addition of impurities on the conductivity of insulator.
- They have very low conductivities ranging between 10–20 to 10–10 ohm–1 m–1.
Semiconductors :
These are the solids with conductivities in the intermediate range from 10–6 to 104 ohm–1 m–1.
The forbidden energy gap between the valence band and conduction band is less than 3 eV. Thus energy gap between the valence band and conduction band is small. At absolute zero, no electrons are available for conduction.
As the temperature increases, many electrons from the valence band may gain external energy to cross the gap between the conduction band and the valence band. Then these electrons will move into the conduction band. At the same time, they will create vacant energy levels in the valence band where other valence electrons can move. Thus the process creates the possibility of conduction due to electrons in conduction band as well as due to vacancies in the valence band. Examples: Silicon, Germanium
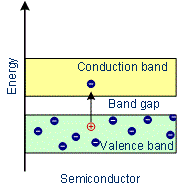
Characteristics of Semiconductors:
- In semiconductors, the conduction band and valence band are very close to each other or the forbidden energy gap between them is very small.
- The electrons of the valence bond can easily be excited to the conduction band.
- There is an energy gap between the conduction band and valence band which is less than 3 eV.
- Due to increase in temperature conductance increases.
- There is an effect of the addition of impurities on the conductivity of semiconductors.
- Their conductivity ranges from 10–6 to 104 ohm–1 m–1.
One reply on “Electrical Properties of Solids”
Very helpful!