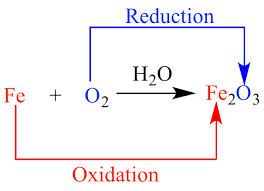
Science > Chemistry > Redox Reactions > Introduction to Redox Reactions In this article we shall study about redox reactions, in which both the oxidation and reduction reactions take place simultaneously. Oxidation: Old Concept: It is a process in which addition of oxygen takes place. 2Mg + O2 → 2MgO It is a […]