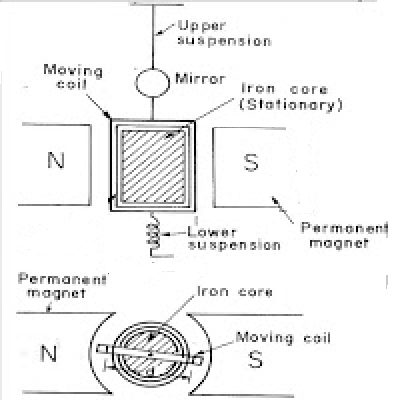
Science > Physics > Magnetic Effect of Electric Current > Moving Coil Galvanometer In this article, we shall study principle, construction, working, sensitivity and accuracy of the moving coil galvanometer Principle: When a current-carrying coil is suspended in a uniform magnetic field it is acted upon by a torque. Under the action of this torque, […]
Moving Coil Galvanometer