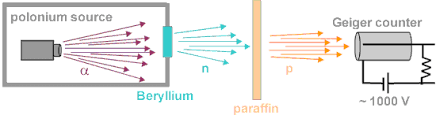
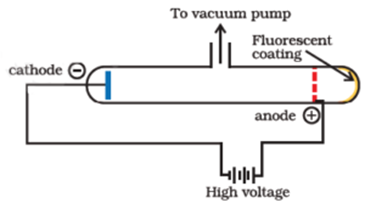
Science > Chemistry > Introduction to Chemistry > Significance of the Study of Chemistry In this article, we shall study the meaning of chemistry and its different branches. What is Chemistry? Science word is derived from the Latin word ‘Scientia’ which means ‘to know’. Science has many disciplines, Chemistry is one of them. It is the […]